A Sprained Ankle Injury Explained
A sprained ankle is one of the most common injuries caused by participation in sports. It refers to soft tissue damage (mainly ligaments) around the ankle, usually caused by an inversion injury (where the ankle is twisted inwards) or an eversion injury (where the ankle is twisted outwards).
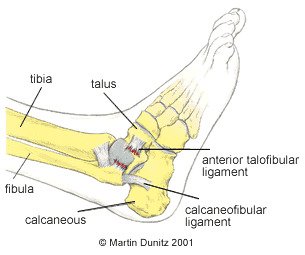
Because of the position of the bones around the ankle, the inversion injury is far more common. This injury causes damage to the lateral ligaments, and other soft tissues, on the outside of the ankle.
The most commonly injured ligament is the anterior talo fibular (ATF) ligament which, as the name suggests, joins the fibular and talus bones together. If the force to the ankle is more severe, the calcaneo fibular ligament (between the calcaneus and fibula) is also damaged. The posterior talo fibular (PTF) ligament is very rarely damaged in comparison to the other two ligaments.
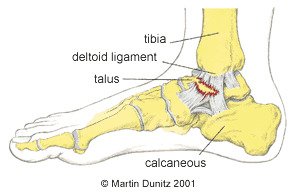
In the case of an eversion injury the damage occurs on the medial (inside) of the ankle. The ligament on the inside of the ankle is called the deltoid ligament and is very strong. It is so strong in fact that the bone on the inside of the ankle can be pulled off, in what is called an avulsion fracture, before the ligament is damaged.
As well as damage to the ligaments, the capsule which surrounds the ankle joint can also be damaged. The damage causes bleeding within the tissues and the ankle begins to swell up and can be extremely painful.
Ankle sprains can be classified as follows:
- First degree, where only a few ligament fibres are damaged
- Second degree sprain refers to more extensive damage to the ligament with associated swelling
- Third degree sprain refers to a complete rupture of the ligament with swelling and a possible joint dislocation
- In the more severe injuries there may be associated bone injury and it is wise to get an x-ray to determine whether there is a fracture.
Sprained Ankle Injury Signs & Symptoms
With a first degree sprain there is pain when turning the foot in or out and also pain when the damaged area is touched. With a second degree sprain the pain is more severe, there is swelling all around the area and it is painful to walk. With a third degree sprain the pain is excruciating and walking is impossible. There is gross swelling and there may be a great degree of joint laxity around the ankle or deformity if the ankle is dislocated.
What you can do
- Consult a sports injury expert
- Apply ice packs/cold therapy to reduce swelling
- Wear a removable plastic cast for protection
- Wear an ankle support for protection
- Wear an ankle support for protection
- Use a wobble board for ankle strengthening
Sprained Ankle Injury Treatment
In the first 48-72 hours following the injury it is important the follow the PRICE protocol – protection, rest, ice, compression and elevation (never apply ice directly to the skin). Ice packs for a period of twenty minutes every couple of hours may help with the pain and help reduce the initial inflammation, but pain-relieving medication may also be necessary. A cryotherapy cuff is the most effective method of providing ice therapy, whilst protecting the injured tissues from further damage, and is the professional’s choice. It can provide continuous ice cold water and compression for 6 hours and significantly reduce pain and swelling in the early stages of injury.
It is important not to put too much weight on the damaged ankle, so walking should be limited where possible. Ankle injuries can be protected using a plastic walker boot cast and these are regularly used by Premier League football players to dissipate weight bearing.
Where a fracture is suspected an x-ray should be carried out at an accident and emergency department. If a fracture is found or a grade three sprain is diagnosed, the advice of the attending doctor should be followed. It should be borne in mind that some hairline fractures do not show up on x-ray until about 10-14 days after the injury, so if the pain persists medical attention should be sought.
In the case of a grade two sprain, crutches should be used to protect the injured ankle. However, it is important not to be on the crutches for longer than necessary and as soon as the pain allows the patient should begin to gently put weight through the ankle by walking to maintain a normal gait pattern and prevent compensatory movements being adopted. Reusable plastic casts can be very helpful as they protect the injured area and help to resolve ankle swelling.
Ligament Damage and Repair Explained
Once the patient is able walk on the ankle, more active rehabilitation can be started.
In ankles that have been repeatedly sprained there is an inherent weakness which may require surgery. This can now be done arthroscopically where a camera is inserted into the ankle and flakes of bone and excess scar tissue can be removed. Because of the injury to vital mechanoreceptors within the ligaments and tendons around the ankle, continuing rehabilitation may be necessary for the months and years that follow an injury. This is because proprioception around the ankle can be greatly compromised with a severe injury and re-injury is often very likely.
Sprained Ankle Rehabilitation
This ankle rehabilitation programme is an example of the progression followed by a professional footballer with a sprained ankle. The principles of rehabilitation apply to all ankle sprain injuries, regardless of the sport. These principles, together with expert input from a healthcare professional can help you recover safely and quickly following a sprained ankle. The progression of the exercises should be based on pain free completion of the previous exercise. All rehabilitation exercises should be preceded by a warm up.
Stage 1: Acute Stage (days 1 to 3)
Follow the PRICE protocol:
Protection
The injured tissues should be protected from further damage. A removable plastic cast is ideal as it helps protect the joint and limit weight bearing. It provides the same protection as a plaster cast but it is lightweight and can be removed by the patient. In the case of Grade 3 ankle sprains crutches should also be used.
Rest
Rest from sports is essential, but even walking on the ankle, if it is painful, may cause further damage and should be avoided. No weight should be put through the injured ankle and crutches should be used. Pain relieving medication prescribed by a doctor may be necessary.
Ice
Ice packs are applied for periods of twenty minutes every couple of hours (never apply ice directly to the skin). The ice packs relieve pain and are thought to reduce bleeding in the damaged tissue. A cryotherapy cuff is the most effective method of providing ice therapy and is the professional’s choice. It can provide continuous ice cold water and compression for 6 hours and significantly reduce pain and swelling.
Compression
A swollen ankle is a sign of a significant ankle sprain. The swelling is caused by bleeding in the tissue and a leaking of cellular fluid secondary to tissue damage. If this swelling is allowed to consolidate it can produce excessive scar tissue and structural changes, which seriously lengthens the rehab period. Compression during the early stages helps to reduce swelling and is essential for a good outcome. Compression is provided by the use of a compression sleeve or brace with intermittent use of a cryo cuff to provide ice and compression.
Elevation
If the ankle is not elevated the effect of gravity causes the tissue fluid to accumulate at the ankle. This can cause increased tissue pressure which is extremely painful. To relieve pain and prevent excessive swelling, the ankle is elevated above waist height.
If the pain allows, the ankle should be removed from the cast and gently pumped forward and back 20 times each hour. This is done by sitting down with the leg elevated and pushing the toes forward and back. This facilitates the dispersal of swelling from the ankle.
Stage 2: Sub-Acute Stage (days 3 to 14)
The sub-acute stage begins by bearing weight on the ankle to pain tolerance. This is graduated from partial weight-bearing with a plastic cast and crutches to full weight-bearing without crutches. A normal walking pattern should be encouraged and there should be no limping.
Ice therapy can be continued so long as there is pain. Compression using the walker boot or ankle brace is continued during walking to encourage the dispersal of swelling. When possible the ankle should be elevated to allow the effect of gravity to help drain swelling away from the injury site. Gentle massage to further encourage the dispersal of swelling towards the back of the knee can also be implemented.
Ankle pumping exercises can be continued and progressed to being done in water. Exercises in water are effective because they involve only partial weight-bearing and because the hydrostatic pressure provided by the water has the effect of encouraging the swelling to disperse.
Exercises in water involve only partial weight-bearing and are effective in helping to disperse swelling.
General fitness is maintained by using a static exercise bike and resistance weight machines in a gym. Resistance Bands may also be used to start to encourage range of movement and gentle strengthening exercises. Different levels of resistance can be used so “light” bands are used at first then as pain, swelling and function all improve the level of resistance can be increased.
Stage 3: Early Rehab (week 3)
After two weeks most of the rehabilitation is achieved through active exercise, although the physiotherapist may help regain range of movement by carrying out massage and passive mobilising techniques. This involves the physio gently moving the bones of the ankle to help restore range of movement and relieve stiffness.
Exercise 1
The first exercise is to help restore the ankles’ range of movement.
Sitting on the floor, the ankle is pumped forward and back.
20 repetitions, 5 times daily.
The patient progresses to doing the same exercise while sitting on a high bench or chair, letting the feet hang down
Exercise 2
This exercise uses a resistance band to strengthen the dorsiflexor muscles that pull the toes back towards the knee.
Sitting on the floor, a resistance band is tied around the foot, with the other end attached to a fixed object in front of the foot.
Slowly the foot is pulled back towards the knee.
20 repetitions, 5 times daily.
As this becomes easier, the resistance band is changed to one that provides more resistance.
Tip: Doubling up the resistance band is an easy way to achieve more resistance.
Exercise 3
This exercise is designed to strengthen the plantar flexor muscles that push the foot down, such as when pushing the pedals of a car.
Sitting on the floor, with the legs out straight, a resistance band is tied around the foot, and the other end held in the hand. Slowly, the foot is pushed forward and then relaxed.
20 repetitions, 5 times daily.
Again, as this becomes easier, the resistance level of the resistance band should be increased.
Exercise 4
This exercise is a progression of the previous plantar flexor strengthening exercises.
The patient stands with their hands resting against a wall so that it is taking some of their body weight. With both feet, the patient pushes up on the toes so the heels rise up off the floor. This position is held for 2 seconds and then the heels are slowly lowered.
20 repetitions, 5 times daily.
Once this becomes easier, the same exercise is done but without any hands against the wall. Once this has been mastered, the patient progresses to doing the exercise with the toes positioned on a block or a step, so that the heels have to come down lower before pushing up.
The final progression to this exercise is to do it on the affected ankle alone.
Exercise 5
This exercise is designed to strengthen the Peroneal muscles that evert the foot and enable the foot to turn outwards. These muscles are also vital for proprioception when returning to sporting activities.
Standing up, the base of the foot is turned outwards, held for 2 seconds and then relaxed.
20 repetitions, 5 times daily.
Proprioception Exercises
Apart from mobilising and strengthening exercises, the rehabilitation for a sprained ankle can be greatly enhanced by practicing what are called proprioception exercises. These enhance neuromuscular control around a joint and are very important to the professional sportsperson, or anybody returning to sports where landing, twisting and turning are necessary. These exercises should be started as soon as pain allows.
The patient balances on the affected leg while partial weight-bearing. This should be done for 1 minute, followed by rest periods to avoid muscular fatigue.
Proprioception exercises can be done while partial weight-bearing first, then progressed to full weight-bearing, such as using a wobble board. The wobble board together with ankle brace are commonly used in the rehabilitation of ankle instability. Wobble boards are designed to assist the re-education of the proprioceptive system by improving sensory nerve function. Research has shown that wobble board training improves single leg stance ability and balance, while other studies have suggested that patients with ankle instability who underwent wobble board training experienced significantly fewer recurrent sprains during a follow-up period than those who did not follow the training programme.
The patient balances on the affected leg for 1 minute, followed by rest periods to avoid muscular fatigue.
Stage 4: Late Rehab (week 4)
Progressive strengthening of the muscles around the ankle should be continued, as should the proprioception exercises. To prepare for a return to functional activities the intensity of exercise should be increased. The use of an ankle brace or ankle taping can provide essential support and confidence at this stage.
Basic plyometric exercises should be commenced.
Jogging should also commence, and should be progressive as follows:
| Day 1 | Jog 100 metres, walk 50 metres, with 6 repetitions. |
| Day 2 | Jog 150 metres, walk 50 metres, with 6 repetitions. |
| Day 3 | Jog 200 metres, walk 50 metres, with 8 repetitions. |
| Day 4 | Jog 200 metres, walk 50 metres, with 12 repetitions. |
| Day 5 | Jog 2000 metres. |
Stage 5: Functional Rehab (week 5+)
The progression to functional activities can begin once the patient can jog without pain and is comfortable doing plyometric drills. The idea of this stage is to progress from gentle exercise to the high intensity at which games are played, by breaking down complex movements and strengthening all of the components. All exercises are preceded by a warm up. As each exercise is a progression they should be completed at least one day apart, under the supervision of a chartered physiotherapist.
Exercise 1
Variable pace running with the gradual introduction of turns.
This involves running round a 20m diameter figure-of-eight course. The figure-of-eight course puts very gentle stress on the ankle and prepares the player for later turning drills. The pace is limited to walk, jog or half pace running and is determined by the physiotherapist who shouts out the desired pace. The physiotherapist also shouts the commands stop and start. This re-introduces the player to the variable demands of a game of football and the Physiotherapist can observe for any compensatory movements that may have resulted from the injury and if not addressed could cause future injuries when returning to activity.
The session should last about 25 minutes.
Exercise 2
Variable pace running with gradual turns and various starting positions.
The player starts at one end of the course and makes a 30m run up to a 20m diameter semicircle, around which they gently turn before completing another straight 30m run back to the finish. The pace of the run is dictated by the physiotherapist and is either a jog or half pace. The starting position should be different for each run (standing, lying on back, lying on front, sprint start position, squatting, right side lying, left side lying, jumping, hopping, facing backwards).
The patient should aim to complete 20 runs.
Exercise 3
A progression of exercise 1 – variable pace running with slightly tighter turns.
Run round a 10m diameter figure-of-eight course. The figure-of-eight course puts stress on the ankle and prepares the player for later turning drills. The paces used are walking, jogging, half pace running, and three-quarter pace running, as determined by the physiotherapist who shouts out the desired pace. The physiotherapist also shouts the commands stop and start.
The session should last about 25 minutes.
Exercise 4
A progression of exercise 2 – variable pace running with gradual turns and various starting positions.
The player starts at one end of the course and makes a 30m run up to a 20m diameter semicircle, around which they gently turn before completing another straight 30m run back to the finish. The pace of the run is either three-quarter or full pace, as dictated by the physiotherapist. The starting position should be different for each run (standing, lying on back, lying on front, sprint start position, squatting, right side lying, left side lying, jumping, hopping, facing backwards).
The player should aim to complete 20 runs
Exercise 5
Two 5m diameter circles are placed 30m apart. Travelling at full pace the player makes a run, with a football at the feet, goes around the far circle and then back to the finish.
This should be repeated 20 times.
Exercise 6
As exercise 5, but single cones are used instead of 5m diameter circles.
Exercise 7
Six cones are placed 5m apart in a straight line. The player completes a shuttle run, at full pace, turning alternately to the left and right.
This should be repeated 10 times
Before a gradual return to full training is considered, the patient should be happy with all normal ball work drills, all types of passing. For instance in football; instep, side foot, front foot, outside of foot, side foot volley, laces volley, half volley, over all distances, heading, jumping and heading, and tackling.
Sprained Ankle Injury Prevention
Ankle instability is characterised by ankle weakness and giving way, even though the ankle ligaments are intact and the joint is mechanically stable. It is due to an impairment of proprioception, which leads to a lack of balance and ankle joint position sense. Proprioception is the mechanism by which nerve receptors in skin, muscle, ligament and joint tissue relay information to the brain about body position sense, where this information is quickly processed and movement strategies are formulated and executed using nerve signals to muscles. This mechanism can help you ‘catch yourself’ when you are about to turn your ankle.
In the unstable ankle these receptors may have been damaged directly during an ankle sprain. The Peroneal muscles and tendons that run along the outside of the shin and are often involved in ankle inversion injuries are particularly rich in receptors. This impaired proprioceptive ability may, therefore, lead to a delay in protective muscle activity and the resultant loss of postural awareness and stability around a joint. This may explain why recurrent ankle sprains are so common.
Wobble Boards together with ankle braces are commonly used in the rehabilitation of ankle instability. Wobble boards are designed to assist the re-education of the proprioceptive system by improving sensory receptor function. Previous research has also shown that wobble board training improves single leg stance ability and balance; while other studies have suggested that patients with ankle instability who underwent wobble board training experienced significantly fewer recurrent sprains during a follow-up period than those who did not follow the training programme. Exercise bands are also useful to help restore muscle strength, especially in multi-directional planes of movement improving the strength and protection around the ankle.
Taping and bracing the ankle can also help to reduce recurrent ankle injury. Previous research has shown the injury incidence in students with taped ankles was 4.9 ankle sprains per 1000 participant games, compared with 2.6 ankle sprains per 1000 participant games in students wearing ankle braces. This compared with 32.8 ankle sprains per 1000 participant games in subjects that had no taping or bracing.